Amphiphiles |
|||||||||||
Aggregates of amphiphile moleculesDownload a printable document of this page in Word (117 KB) or pdf (273 (KB) format.
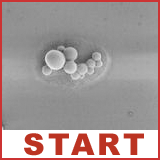
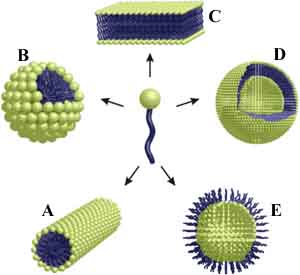
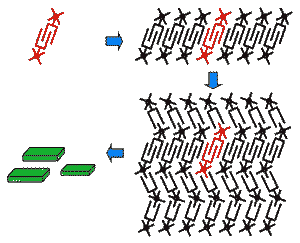
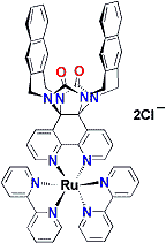
Hydrophilic (hydros = water; philic = loving) molecules dissolve well in water (a polar solvent), whereas hydrophobic (waterminding) molecules dissolve best in an apolar solvent. Molecules that consist of both a hydrophobic and a hydrophilic part are called amphiphiles (amphi = both). Amphiphile molecules orientate their domain with the highest affinity for the solvent toward that solvent, while the other part of the molecule turns to avoid contact with the solvent. In this way amphiphilic molecules align according to well-defined patterns and give rise to pluriform aggregates. The shape (=morphology) of the aggregates is determined by the ratio between the size of the hydrophilic and the hydrophobic part as well as the nature of the solvent. Scanning electron microscopy and other techniques have revealed various morphologies: micellar rods, micelles, bilayers, vesicles and inverted aggregates. The washing property of soap is based on amphiphiles that interfere between water and (fatty) dirt. Amphiphiles also form the main part of the cell membranes that serves to delimit and protect cellular compartments. Spherical aggregates of polystyrene-block-polyisocyanides: "microcontainers"The bead-like aggregates shown in this demonstration are made of polystyrene polyisocyanides (abbreviated PS-PIAT; see formula) that have been synthetized at the Department of Organic Chemistry (Radboud Universiteit Nijmegen). The spherical aggregates have been obtained by dissolving the PS-PIAT polymer in an organic solvent (tetrahydrofuran) and injecting the mixture into water. The aggregates resemble vesicles, since they consist of a membrane bilayer sheath and are hollow in the centre. The vesicles have a diameter of about 1.5 micrometer, are very stable and retain their shape after air-drying. It was demonstrated that dye molecules and even enzymes could be included inside these vesicles. The micro-vesicles system is particularly interesting, because it offers perspectives for applications as micro/nanoreactor.Self-assembling artifical receptors: "molecular clips"Other amphiphiles that are synthesized and studied at the departments of Organic Chemistry and Solid State Physics 2 are the so-called "molecular clips". They are artifical receptors that consist of a U-shaped hydrophobic cavity and a hydrophilic ruthenium tris(bipyridine) complex (structure formula left). In water, these molecules self-assemble by filling up each other's cavities (central diagram). Eventually, rectangular aggregates are formed (fesem image right; scientist: H. Elemans), which contain thousands of molecules and which all have a well-defined shape and a similar size.
The ordering of the molecules was confirmed by X-ray powder diffraction and high resolution Nuclear Magnetic Resonance techniques. The arrangement of the chromophoric ruthenium complexes renders these aggregates interesting for further development as photoactive elements.
(More information on this topic and nanotechnology: J.A.A.W. Elemans, A.E. Rowan, and R.J.M. Nolte, J. Am. Chem. Soc. 2002, 124, 1532-1540). Software development: Jeroen van Beurden. Web structure: Remco Aalbers. Text and images: Dennis Vriezema, Hans Elemans, Elisabeth Pierson and Huub Geurts |
|||||||||||